This week’s post was written with the help of Dr Jorn Cheney, post-doctoral fellow at the Royal Veterinary College. If you would like to contribute a guest post, please get in touch on Twitter or Facebook.
Powered flight has evolved four times i n the animal kingdom: once each in insects, birds, pterosaurs and bats. Biomechanists have spent decades examining the anatomy, physics and energetics of flight in insects and birds, the largest of these two groups, and this has led to the application of biological principles in aeronautics and engineering. In these cases, the wing is a firm, rigid structure whose shape is fine-tuned to suit its purpose, maximising lift (forces keeping bats aloft) and reducing drag (forces slowing bats down). However, bats have taken an entirely different approach to wing design: their wings are thin, delicate and highly flexible, being supported only by the elongated bones of the hand. The membrane stretching across the span of the wing forms the aerodynamic surface that powers it.
n the animal kingdom: once each in insects, birds, pterosaurs and bats. Biomechanists have spent decades examining the anatomy, physics and energetics of flight in insects and birds, the largest of these two groups, and this has led to the application of biological principles in aeronautics and engineering. In these cases, the wing is a firm, rigid structure whose shape is fine-tuned to suit its purpose, maximising lift (forces keeping bats aloft) and reducing drag (forces slowing bats down). However, bats have taken an entirely different approach to wing design: their wings are thin, delicate and highly flexible, being supported only by the elongated bones of the hand. The membrane stretching across the span of the wing forms the aerodynamic surface that powers it.
Initially, this seems a passive and even clumsy design for a wing: it is controlled by surprisingly few bones and muscles, and the membrane shape would be almost entirely defined by the movement (or not) of air around it. But it is clear from observations of bats in flight that they are highly powerful, dextrous flyers capable of fine and complex manoeuvres (see video). So how do they achieve this? Dr Jorn Cheney, a new post-doctoral researcher at the RVC, worked on this question for his PhD and post-doc at Brown University in Rhode Island, USA.
Cheney’s first step was to look more closely at the structure of the skin that forms the wing membrane. When unloaded, or when the bat is not in flight, the membrane is gathered and wrinkled in appearance, stretching out to taught arches during wingbeats. Dissections reveal two unusual features of the wing membrane that stood out in particular: the skin contains both bundles of elastin (a springy, elastic protein) and long, fine bundles of skeletal muscle. Although it’s normal for skin to contain both muscle and elastin fibres, these bundles are much larger in bat wing membranes and their function is unclear.
By mapping them across the wings of several species using polarised light, the group found that the elastin bundles are arranged almost in parallel across the span of the wing. They also found that by very carefully removing these fibres, the neat wrinkling of the wing disappeared. Cheney and his

colleagues believe the elastin fibres act in the same way as a pleated elastic waistband, organising the membrane when compacted, and allowing it to expand neatly in a certain direction when necessary. The fibres are already under a little strain when the wing is relaxed, pulling the membrane into its wrinkled formation, but when the wing expands the elastin takes the load, rather than the rest of the membrane tissue, and the wrinkles are smoothed out.
The muscles, on the other hand, extend across the wing in the opposite direction. Cheney and his colleagues have now traced these plagiopatagiales proprii (snappy name) and documented their patterning in 17 of the 18 families of living bats (here are two examples below, with beautiful illustrations from a student working on the project, Elissa Johnson). Previous work had suggested that the muscles might primarily act as stretch receptors in the wing. However, by monitoring the activity of the muscles through attached electrodes, the team discovered that they are activated at the top of the upstroke through to the end of the downstroke and, remarkably, activate simultaneously. This synchrony allows this array of small, weaker muscles to create a considerable total effect, stiffening the wing and reducing membrane deformation during the all-important downstroke which powers flight.

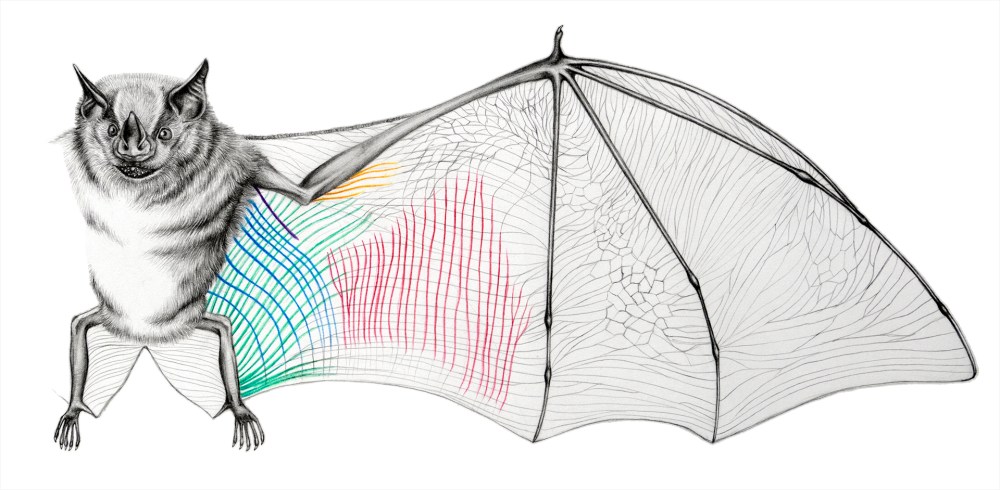
Together, these two adaptations allow the bats unprecedented mechanical control over the shape and structure of the wing membrane: the passive yet effective elasticity of the wing and the active muscular control over wing stiffness. As we can see from the illustrations above, the precise arrangement of these networks varies considerably between species. Given the enormous diversity of lifestyles found in the 1,200 species of there is an exciting possibility that these differences might have something to do with it, affecting their wing shape and so potentially their speed, manoeuvrability and other aspects of flight. The sophistication of the system discovered by Cheney and colleagues may also prove invaluable to the development of fight technology, but will certainly provide a fascinating research area for years to come as they delve further into the aeronautic ability of these fantastic animals.
Images courtesy of Jorn Cheney. Illustrations accredited to Elissa Johnson. To see more of Elissa’s work, see her blog here. To find about more about bats in flight, see the group’s site (at Brown.edu).
Glossary:
Plagiopatagiales: Muscles embedded within the wing membrane (non-specific).
Plagiopatagiales proprii: A group of long, slender muscles which run along the wing membrane parallel to the main body.