Our guest post this week comes from Haley O’Brien, a PhD student at Ohio University, and Dr. Jason Bourke from the North Carolina Museum of Natural Sciences. If you would like to contribute a guest post, please get in touch on Twitter or Facebook.
Both giraffes and sauropod dinosaurs are renowned for their signature long necks. As scientists these super-long necks intrigue us because they seem to create a host of physiological problems that nature has had to solve. For instance, have you ever noticed that when you stand up too fast you can sometimes feel dizzy?

This is because our circulatory system, which must fight against the constant pull of gravity, gets strained by our brain’s sudden change in height. If you’re a giraffe, this height problem is increased by 2 meters (6.5 feet), and this distance would be even greater for many sauropod dinosaurs. For a long time, it was thought large arterial meshwork at the base of the giraffe brain, called the carotid rete, helped giraffes solve two major problems associated with their long necks: preventing them from fainting when they raised their heads, and keeping their brains from suffering catastrophic pressure increases caused by blood rushing to the brain when lowering their heads to drink. The proposed use of the carotid rete as a pressure-absorbing capacitor for giraffes led other researchers to suggest that sauropods may have employed similar structures to help protect their brains, too.
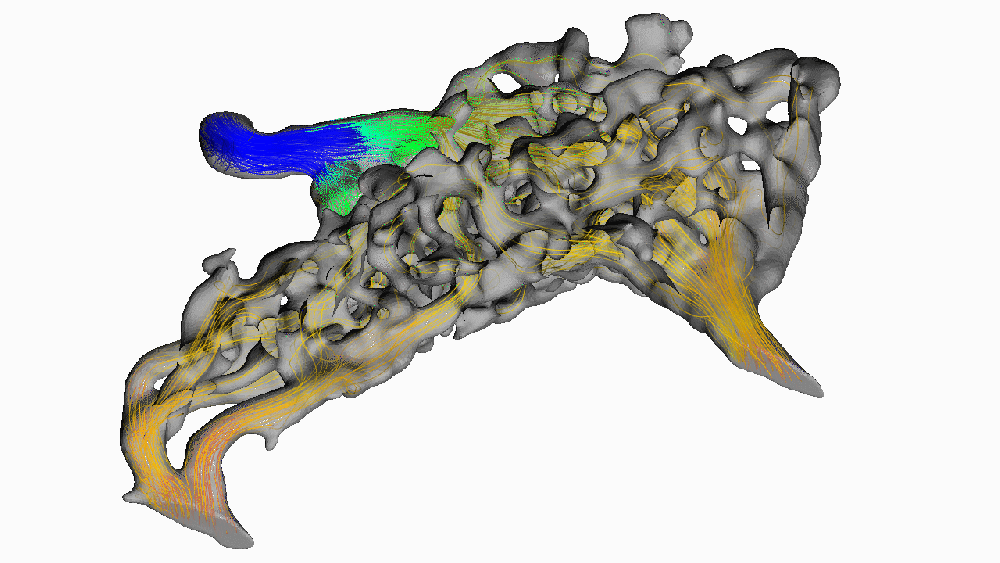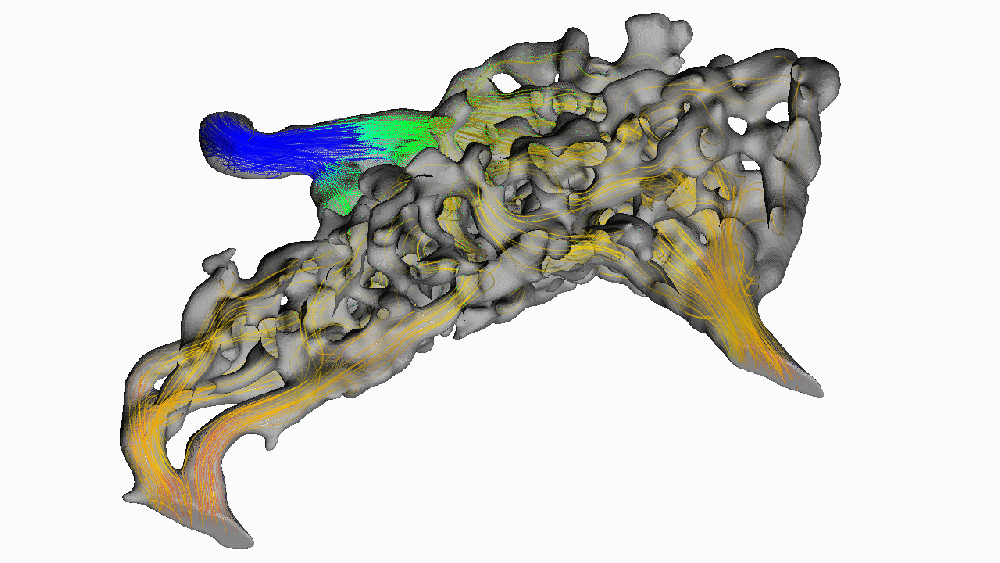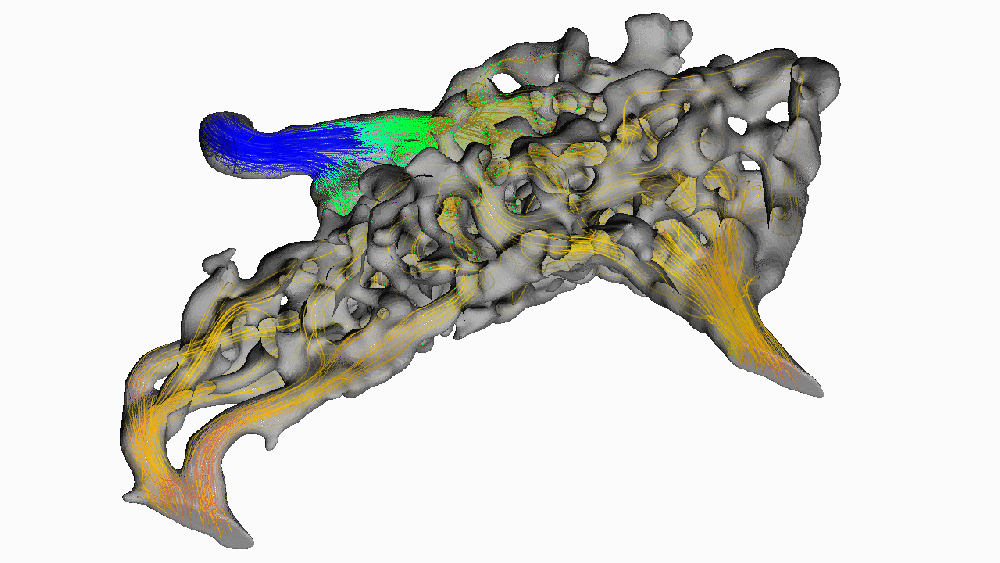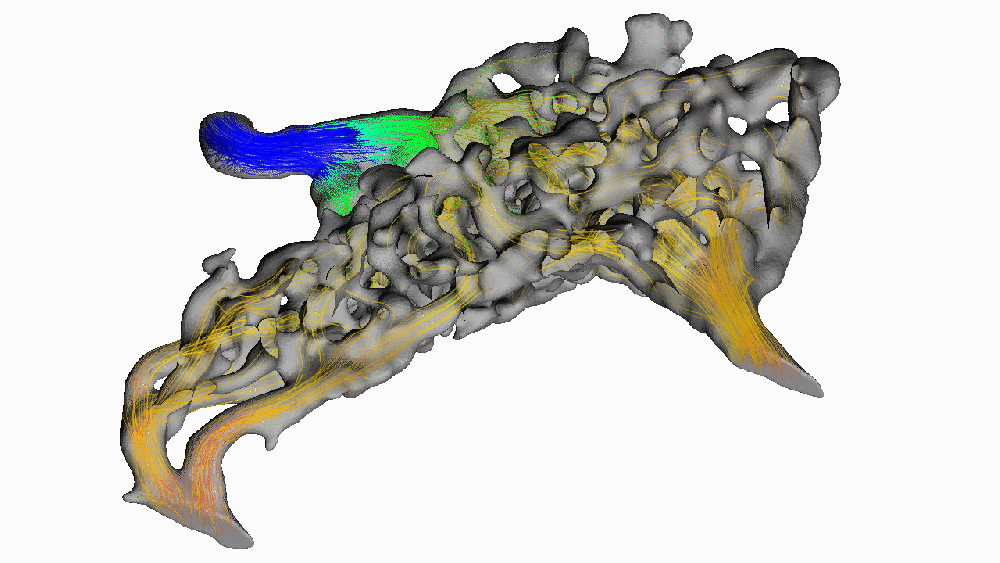
Despite its popularity, this hypothesis has been difficult to test in living animals. Recently, we used new digital 3-D modeling techniques to simulate what happens to blood as it flows through the carotid rete. Using a method called computational fluid dynamics, we digitally “pumped” blood through the rete under normal, head-raising, and head-lowering scenarios. We found that this arterial structure had essentially no impact on blood flow. The image below shows blood pressure change throughout the rete during the highest pressure phase. Warm colors indicate high pressure, and cooler colors indicate lower pressure.
Notice how almost all of the arterial meshwork is the same shade of green? (moderately low pressure) We found that pressure dropped by a measly 1.5 mmHg (around 0.002 atmospheres) before entering the brain. Given that typical blood pressure for a giraffe when drinking is about 330 mmHg (0.45 atm), a change of 0.5% doesn’t mean much for protecting the animal’s brain. We found this result surprising since it goes against the long-standing conventional wisdom that giraffes use their carotid rete to mitigate blood flow to their brains.
So then how do giraffes and sauropods keep themselves from fainting every time they raise their head to feed, or avoid brain bleeds when bending down to drink water?
Unfortunately, we’re still not sure. However, by testing this hypothesis, we’ve opened the door for others to test alternative processes, such as diverting blood into the jugular or vertebral veins or shunting blood away from the brain into arteries that don’t enter the braincase. Digital modeling helped us see what doesn’t work; now we can start looking for what does.
Very interesting! But can you please add a link to the paper.
How confident are you that the modelling reflects reality? Have you been able to test it against observed pressure effects in live animals? I ask because I have become quite cynical about certain engineering techniques’ application to animals.
LikeLike
Whoopsie; link added! (http://www.sciencedirect.com/science/article/pii/S0022519315004518)
LikeLike
Thanks!
LikeLike
I agree with your cynicism about applying engineering techniques to biological systems, and I think it’s important to understand the context and limitations of the analyses to insure you’re using them appropriately. But that said, we’re pretty confident for a number of reasons that are unique to the modeling methods we used.
First off, we used two different models, and they both came up with congruent results. The first model is a straight-forward application of Poiseuille’s equation which physicians have used for decades to broadly assess cardiovascular risk. It’s basic pencil-and-paper physics/hemodynamics that’s known to be accurate enough to diagnose and treat humans. The computational fluid dynamics method that we used has a huge volume of literature assessing its accuracy in both human and animal models, including direct comparisons between the CFD models and in-vivo measurements. Again, it really helps that these vascular models have a medical application where inaccuracies could be seriously detrimental to human health.
On top of that, our input blood pressures were taken directly from measurements we found in literature, and the output pressures where the retial segments coalesce to form the intracranial internal carotid artery match those measured at the middle cerebral artery a bit further downstream. There’s a lot of data on goats and pigs in particular, because they’re frequently used as medical models. So in other, non-giraffe artiodactyls we have great measurements of pressure upstream and downstream from the rete, but the meshwork itself has always been a mystery.
And finally, the histology and receptor physiology of the rete corroborate the models as well. The up- and downstream vessels are highly contractile, but the walls of the retial segments aren’t particularly muscular and they almost completely lack a response to adrenergic stimulation. The rete isn’t contractile, so it wouldn’t be particularly able to deform or constrict to control blood pressure either.
So ultimately, we’re confident that these particular engineering principles can be accurately applied to this system. I hope that answers your question!
LikeLike
Hi Mike,
You are right to be skeptical of digital models for animals. The fact that digital models have the capacity to replicate any scenario thrown at them makes them both a gift and a curse. We always kept this in mind during our analysis and strove to make sure that models were given appropriate physical constraints based on the biological reality of the organisms. We took data from empirical studies whenever we could (e.g., systolic and diastolic pressure values at both the heart and maxillary artery, blood pressure when standing vs. recumbent, etc.) and we made a point of couching all of our inferences within the realms of biological feasibility.
For instance, our carotid rete was modeled as a static structure, incapable of stretching or contracting. For a blood vessel this would typically be an unrealistic assumption to make, but extensive work on the physiology and histology of artiodactyl carotid retia have shown that it is an almost rigid structure (e.g., Dieguez et al. 1987), so we felt justified in this modeling assumption.
There are some data out there on carotid rete pressures, albeit not for giraffes. Studies such as Lluch et al. (1985) and Dieguez et al. (1988) on goats and other domestic artiodactyl retia (which are near identical to a giraffe’s) found little to no pressure changes across the rete during standing and resting, along with a near total lack of response to vasoconstrictive or vasodlating hormones. Barring some unique anatomy to giraffe carotid retia, we think our models show a fairly accurate representation of blood pressure change across this structure.
References
Diéguez, G., García, A.L., Conde, M.V., Gómez, B., Santamaría, L., Lluch, S., 1987.
In vitro studies of the carotid rete mirabile of Artiodactyla. Microvasc. Res. 33,
143–154.
Diéguez, G., García-Villalón, A.L., Gómez, B., Lluch, S., 1988. Hemodynamic significance of the carotid rete during changes in arterial blood pressure. Am. J.
Physiol. 254 (5 Pt. 2), 770–775.
Lluch, S., Diéguez, G., García, A. L., Gómez, B. 1985. Rete mirabile of goat: Its flow damping effect on cerebral circulation. Amer. J. Physiol. 249:R482–R489.
LikeLike
Thanks, Jason — that’s very helpful, and compelling.
LikeLike